Renowned Speakers

Sara Torgal
Senior Manager, Scientific Programs DIA Swaziland

Bandar Al Hammad
Chief Pharmacist, Regulatory Affairs Department Saudi Food and Drug Authority Saudi Arabia

Amira Deia Younes
Associate Director, Reg Policy& Intelligence MEA, Russia, CEE & EFPIA MERN Chair UAE

Fadila Lakkis
Regulatory Affairs, Intelligence & Communications Manager, Gulf GSK, United Arab Emirates UAE

Inas Chehimi
Head RA Middle East & North Africa Novartis Pharma Services AG, United Arab Emirates UAE

Catherine Al Ashram
Executive Director Regulatory Affairs & PV Lead, EEMEA Organon, Jordan Jordan

Amani Mansour, MSc
Gulf Regulatory Affairs Working Group Co-Chair & Head of Regulatory Affairs-Gulf UAE

Samia Gamal Seleem,
Scientific & Regulatory Affairs Director Abbvie, Egypt Egypt
Recommended Global Pharmaceutical Sciences Webinars & Conferences
Asia Pacific & Middle East
Pharma Expo-2023
- About Conference
- Scientific Sessions
- Participation / Presentation Option
- Abstract Submission Guide lines
- How to complete registration?
- VISA Requirements
- Market Analysis
About Conference
The most anticipated Pharma event in the world and you're invited to attend 22nd International Pharmaceutical Sciences & Life Saving Drugs IPSLSD-2023 scheduled during August 28-29, 2023 at Alberta, Canada. Which includes prompt keynote presentations, Oral talks, Poster presentations and Exhibitions? This strategic conference will provide in-depth presentations and interactive sessions, for the possibility to network with like-minded professionals and discuss how to overcome the current challenges in pharmaceutical & Drugs product synthesis and product development. The focus this year will be on the latest advancements, research and development to inspect the challenges in Pharmaceutical Sciences & Life Saving Drugs and related areas. LIFE SAVING DRUGS-2023 IPSLSD-2023 is a specially designed cluster of all Pharma conferences. The main theme of this Pharma conference is "Outlining the most recent developments in pharmaceutical sciences and life-saving medications" which covers a wide range of critically important sessions.
At LIFE SAVING DRUGS-2023 meet your target audiences from around the world focused on learning about Pharma and Drug regulations, licenses, and approvals. This conference would be your single best opportunity to reach the largest assemblage of participants from the Pharma field.
2023 Highlights:
300+ Participation (70 Industries: 30 Academia)
10+ Keynote Speakers
50+ Plenary Speakers
20+ Exhibitors
14 Innovative Educational Sessions
5+ Workshops
B2B Meetings
Motives to attend:
This year, we will have more senior speakers, who will be joining us from IPSLSD-2023 pharmaceutical giants. They're coming to share their experience with us and ensure you can maximize your knowledge. They also want to join us because they know how important it is to have your questions answered. In light of all that's happening in the Pharmaceutical industry, this year delivers more case studies for you to gain first-hand experience on developing your drugs, and we will be introducing think tank sessions to discuss and debate challenges in more detail with key decision makers. These sessions will bring you constructive networking possibilities so you return to work with practical solutions for your challenges. Get ahead of the competition: Gather fresh intuitions from key speakers to help make restored decisions on doing business in Pharma Meet & Connect with Senior Stakeholders from the global Pharma network
Content your way: Access a variety of technical and commercial tracks across the entire supply chain, or choose the one track that is most relevant to your business Save months of research: Don’t spend eras reading whitepapers, come and hear from the decision makers themselves on the latest trends, challenges and opportunities at the forefront of the industry A Unique Opportunity for Advertisers and Sponsors at this International event
Triumph of Awards, Certificates recognizes your commitment to your profession to encourage the nascent research.
Over 50+ organizations and international pavilions will be exhibiting at the LIFE SAVING DRUGS-2023 IPSLSD-2023 conference. Exhibitors will include equipment manufacturers and suppliers, systems providers, finance and investment firms, R&D companies, project developers, trade associations, and government agencies.
In addition to the products and services you will have access to valuable content, including Keynote Presentations, Product Demonstrations and Educational Sessions from today’s industry leaders.
Target Audience:
Directors, CEO’s of Organizations
Business Development Managers
Chief Scientific Officers
R&D Researchers from Pharma Industries
Professors, Associate Professors, Assistant Professors
PhD Scholars
Patent Attorneys
Investment Analysts
Association, Association presidents and professionals
Noble laureates in Health Care and Medicine
Bio instruments Professionals
Bio-informatics Professionals
Software development companies
Research Institutes and members
Supply Chain companies
Manufacturing Companies
CRO and DATA management Companies
Training Institutes
Business Entrepreneurs
Scientific Sessions
Track 1: Pharmaceutical Sciences: Academic and Industry Overview
The pharmaceutical industry discovers, develops, produces, and requests medicines or pharmaceutical medicines for use as specifics to be administered to cases (or tone- administered), with the aim to cure them, vaccinate them, or palliate symptoms. A pharmaceutical science combines with a Wide range of scientific disciplines that involves with the designing, delivery, action, disposition and use of medicines. This field connected with numerous areas of the introductory and applied sciences similar as chemistry, biology, epidemiology, statistics, chemo criteria, mathematics, drugs and chemical engineering and applies their principles to the study of drugs. Pharmaceutical science, defined by the AACP Research and Graduate Affairs Committee, encompasses a broad range of interdisciplinary fields related to drug discovery, optimization, delivery, optimal dosing, as well as health issues and policy.
Track 2: Pharmacognosy and Photochemistry
Pharmacognosy is defined as “the study of the physical, chemical, biochemical and natural parcels of drugs, drug substances, or implicit drugs or drug substances of natural origin as well as the hunt for new drugs from natural sources.” Photochemistry is the branch of chemistry concerned with the chemical goods of light. Generally, this term is used to describe a chemical response caused by immersion of ultraviolet, visible light or infrared radiation. The branch of Science concerned with medicinal drugs attained from plants or other natural sources Pharmacognosy is in the strict feeling of the word came after disquisition of phytochemicals. A significant number of these are known not security against bug assaults and plant affections.
Track 3: Pharmaceutical chemistry
Pharmaceutical chemistry is the study of drugs, and it involves medicine development. This includes medicine discovery, delivery, immersion, metabolism, and more. There are rudiments of biomedical analysis, pharmacology, pharmacokinetics, and pharmacodynamics. Pharmaceutical chemistry work is generally done in a lab setting. The end is to gain new chemical motes that could enable the discovery of new pharmaceuticals or optimize formerly known medicine structures, thereby to expand the portfolio of chemical medicines.
Track 4: Public Health
Public health is the science of protecting and improving the health of people and their communities. This work is achieved by promoting healthy lives, probing disease and injury forestallment, and detecting, precluding and responding to contagious conditions. Public health promotes the weal of the entire population, ensures its security and protects it from the spread of contagious disease and environmental hazards, and helps to insure access to safe and quality care to profit the population. Public health works to track disease outbreaks, help injuries and exfoliate light on why some of us are more likely to suffer from poor health than others. The numerous angles of public health include speaking out for laws that promote smoke-free indoor air and seatbelts, spreading the word about ways to stay healthy and giving science- grounded results to problems.
Track 5: Industrial and Physical Pharmacy
Industrial pharmacy is a discipline which includes manufacturing, development, marketing and distribution of medicine products including quality assurance of these conditioning. Industrial Pharmacy deals with studying all the unit operations involved in medicine delivery manufacturing, the scaling up, processing and development of these drug delivery systems and lozenge forms at the artificial position and encompassing quality assurance with its branches quality control and Good Manufacturing Practice. Physical pharmacy is the branch of pharmacy that concentrates on the operations of drugs and chemistry to the study of pharmacy. In other words, it's the study of the goods that lozenge forms have on their terrain by addressing issues at the molecular position. It emphasis on the physical characteristics and conduct of the drug delivery system before the same is given to the patient.
Track 6: Pharmaceutical Manufacturing
Pharmaceutical manufacturing is the process of industrial- scale conflation of pharmaceutical medicines as part of the pharmaceutical assiduity. The process of medicine manufacturing can be broken down into a series of unit operations, similar as milling, granulation, coating, tablet pressing, and others. The pharmaceutical assiduity includes the manufacture, birth, processing, sanctification, and packaging of chemical accoutrements to be used as specifics for humans or creatures. The Pharmaceutical Manufacturing Technology (PMT) program is a technical chemical technology program designed to meet the requirements of the medicinal and related industries (cosmetics, food, plastics, custom chemicals, exploration centers, pilot plants etc.).
Track 7: Pharmaceutical Analysis
Pharmaceutical analysis is mainly concentrated in drug analyses, in raw materials and pharmaceutical phrasings, involving the determination of active factors, contaminations, excipients, content uniformity, solubility, dissolution rate and stability. The purpose of Pharmaceutical Analysis is to identify substances, purify them, separate them, quantify them, determine the molecular structures of chemical composites that make up medicinal, and determine how these compounds are combined to make up a pharmaceutical product. The HTS technique has been used extensively in drug design and discovery for identification, quantification, characterization of chemical composites. By applying the HTS styles, the remedial, metabolic, pharmacokinetic, toxicological and analytical and degradation data of new medicines can be attained.
Track 8: Nano medicine and Biomedical Application
Nano materials have been extensively used in drug and medicinal because of their specific mechanical, optic and electrical actions. Nano materials are applied for the discovery of natural motes, imaging of diseased tissues and innovative therapeutics. Research in Nano medicine spans a multitude of areas, including medicine delivery, vaccine development, antibacterial, opinion and imaging tools, wearable bias, implants, high- outturn screening platforms, etc. using natural, biological , biomimetic, or cold-blooded accoutrements . Biomedical applications frequently involve the integration of a synthetic device, whether for interrogation or manipulation, with a living, natural organism.
Track 9: Drug Discovery, Design & Development: Challenges and Approaches
The drug discovery process has seen numerous advancements over the decades. New technology has been a crucial motorist of similar advances, with improvements in assay technology, robotization, imaging, Nano fluidic, and software helping to significantly ameliorate the process of drug discovery. Drug development and discovery includes preclinical exploration on cell- grounded and animal models and clinical trials on humans, and eventually move forward to the step of carrying nonsupervisory blessing in order to vend the drug. Maybe the most burning issue in health care presently is the high cost of care. Further than 45 of American grown-ups say it's delicate to go health care, according to a check by the Kaiser Family Foundation, and further than 40 have medical debt.
PRECLINICAL TRIALS: Preclinical trial- a laboratory test of a new drug or a new medical device, generally done on animal subjects, to see if the hoped- for treatment really works and if it's safe to test on humans.
Track 10: Novel Drug Delivery System
Novel drugs, also referred to as new molecular entities or new compounds, are realities that are different from any preliminarily- approved United States Food and Drug Administration (FDA) drug product. These drugs are innovative products that offer a new system of treating certain medical conditions. Novel drug delivery system( NDDS) is an expression substantially associated with the expression of new pharmaceutical forms which have optimized characteristics similar as lower flyspeck size, advanced permeability parameters, and selective site targeting. In the form of a Novel Drug Delivery System a being drug patch can get a new life. A designed Novel Drug Delivery System can be a major advance for working the problems related towards the release of the drug at specific point with specific rate.
Track 11: Pharmaceutical Microbiology
Pharmaceutical microbiology is a specialist area of microbiology and one concerned with the use of microorganisms in pharmaceutical development and with maintaining impurity control. People trained in pharmaceutical microbiology, frequently known as pharmaceutical microbiologists, substantially work in quality control and assurance and department in pharmaceutical companies, and their primary part is to insure the quality of raw accoutrements before they're reused in the production area. Arbro's pharmaceutical microbiology testing ensures that the raw materials used in drugs match the standard quality conditions before they're reused in the product environment.
Track 12: Pharmaceutical Nanotechnology
Pharmaceutical nanotechnology is the exciting, fleetly arising branch of medical science that deals with employing Nano scale accoutrements as medicine delivery and/ or individual tools. As drug delivery tools, Nano- delivery systems can be used to enhance the point-specific, targeted delivery of precise drugs. Nanoparticles can be made by altering the system of fabrication. Nanoparticles have been proven to be useful as drug delivery vehicles. Numerous uses for Nano particulate drug delivery systems live, including gene remedy, cancer therapy, AIDS remedy, and radiation
Track 13: Pharmaceutical Care and Health System
The principal elements of pharmaceutical care are that its drug related it's care that's directly handed to the patient it's handed to produce definite issues these out- comes are intended to ameliorate the case's quality of life and the provider accepts particular responsibility for the issues. Pharmacist, working in collaboration with other health care providers, takes over responsibility for patient issues with respect to their medicine remedy. The top task of the ultramodern pharmacist is to identify, resolve and help drug- related problems. The health requirements of a population cannot be met without these medical products, and the medicinal systems that regulate, force, and promote their applicable use. Strong pharmaceutical systems are necessary for icing high- quality health care, particularly for the most-vulnerable populations.
Track 14: Advanced Pharmaceutical Technology
Pharmaceutical technology is one of the pharmaceutical sciences dealing with the composition, expression, medication or manufacturing and quality control of extemporaneously compounded or commercially manufactured medicines. Pharmaceutical technology is a field of drug which deals with medication, dosage and side goods of different drugs. The course provides the scholars introductory knowledge on the manufacturing process and different machines used in the product of the tablets of any drug.
The top 5 Medical Innovations
Next Generation of mRNA Vaccinology
PSMA- Targeted remedy in Prostate Cancer
New Treatment for the Reduction of LDL
Novel Drug for Treatment of Type 2 Diabetes
Advance Treatment for Postpartum Depression
Track 15: Pharmaceutical Sciences
Pharmaceutical science is the study of the expression, development and manufacture of new drugs. Pharmaceutical scientists have the power to transfigure cases' lives by developing new treatments for conditions. Pharmaceutical science is a field that focuses on the design and conflation of implicit drugs, as well as their evaluation in natural systems and complaint models as a means of profiling their preclinical safety, efficacy, dynamics, and metabolism.
Participation / Presentation Option
Oral Presentation: Oral presentations may include research, theoretical, professional or private practice topics in a concise manner. People with personal experience are also encouraged to submit personal experiences or stories that help others in daily life.
Speakers with a 30-minute slot should plan to speak for 20-25 minutes, and keynote speakers should plan to speak for 40-45 minutes, with the remaining time to be used for questions and discussion by the chair of the meeting session.
Workshop: For workshop presenters as well, the lecture topic will be the same as an oral presentation with more specialized techniques and detailed demonstration. The general duration of a workshop presentation is approximately 45-50 minutes. Interested participants can join their respective team and present the workshop with their research coordinators with special group waivers upon registration.
Poster Presentation: The Student Poster Competition will be held during the IPSLSD-2023 conference to encourage students and recent graduates to present their original research. Presenters will have approximately 5-7 minutes to present the poster, including questions and answers. Judges may ask questions when judging the presentation. This is an opportunity for young scientists to learn about recent discoveries from their peers in order to increase their capacity as multidisciplinary researchers. Poster displays will be in 1x1M long paper size.
For more details on poster presentation and judging criteria, see the poster presentation guidelines.
Webinar: The webinar presentation is designed for interested attendees who cannot attend in person due to scheduling conflicts or other obligations. In this option, the presenter can record the presentation and their presentation will be shown in the webinar presentation session.
E-Poster: E-Poster is also like the webinar presentation. At this session, their presentation will be published as a poster on the conference website and the presenter's abstract will be published in the conference memento and in the journal with DOI.
Exhibit: IPSLSD-2023 may exhibit the products and services of commercial and non-commercial organizations such as artificial intelligence, digital talent, platform-based software and services, trade commissioners, and equipment manufacturers.
To learn more about exhibitor booth details and benefits, visit WHY EXHIBIT WITH US?
Send your questions to lifesavingdrugs@asiapacificmeets.com
Advertisement: The conference program is a valuable resource that all attendees refer to again and again as they navigate the conference. Advertising in the conference program is a great way to market and can help secure long-term business.
Send your proposal to lifesavingdrugs@asiapacificmeets.com for available ad options and pricing
Sponsor Life Saving Drug-2023
Premium Referral Pack
Additional sponsorship package
Email the Program Manager at lifesavingdrugs@asiapacificmeets.com to inquire about sponsorship packages.
Abstract Submission Guide lines
How to submit an abstract/article?
Abstract submissions are welcoming you all to submit abstracts on Life Saving Drug Topics. Please see below for submission guidelines.
Full abstracts accepted for registered attendees will be published in the open-access conference support journal and conference memorabilia.
Submit your abstracts via the online ABSTRACT SUBMISSION link
Abstracts should be submitted in English and should be between 250 and 300 words, along with a 60-70 word presenter biography.
Submissions are welcome on all topics related to Life Saving Drugs & Pharmaceutical Sciences.
The presenting author should provide the following details: presenter's full name, affiliation details: department, institution/hospital, country, phone number, email address, and photograph
For co-authors, the following details are required: full names and affiliation details such as the name of the department, institute/organization, and country should be included
All abstracts will be reviewed by a review board approved by the Life Saving Drugs-2023 conference review board. The Committee's decision is final.
All accepted abstract presenters will be required to register and book a slot to attend the conference.
Please note that your affiliation and abstract title in the final program and in the journal will appear exactly as you submit them. Please, therefore, follow the guidelines provided as to the required format and the letter of acceptance does not imply any travel subsidy or scholarship.
Next steps
An email confirmation will be sent to you after the successful acceptance of the abstract by organizing committee members. If the abstract is accepted, presenters must confirm the presenter(s) and book a slot to confirm their presence at the conference. Failure to do so may result in your paper being excluded from the Life Saving Drug-2023 conference program.
How to complete registration?
To complete registration, visit the ONLINE REGISTRATION PAGE where the following registration categories are available
Entrants from academia/academic backgrounds must register their entry under the academic category
Registration options available in this category: Oral presentation | Poster presentation | Registration of delegates | Package A (includes your registration and 2 nights’ accommodation) | Package B (includes your registration and 3 nights’ accommodation) | Online seminarThe businessman must register his participation in the Business category
Registration options available in this category: Speaker Registration | Registration of delegates | Package A (includes your registration and 2 nights’ accommodation) | Package B (includes your registration and 3 nights’ accommodation) | Advertising | Exhibitor
Participants who are still pursuing their studies will register their participation in the student category.
Registration options available in this category: Speaker Registration | Poster presentation | Registration of delegates | Package (includes registration and 2 nights’ accommodation).
Apart from any category mentioned above, please email lifesavingdrugs@asiapacificmeets.com for GROUP REGISTRATION, EXTRA NIGHT ACCOMMODATION, ADVANCE PAYMENT OF TOKEN, MORE THAN ONE PRESENTATION, etc. These options are available upon special request and discussion with management.
VISA Requirements
The Life Saving Drug-2023 Organizing Committee hereby reiterates that we are NOT involved in VISA processing. We can help you by providing supporting documents such as an invitation letter, abstract acceptance letter, and registration payment receipt.
You may need to submit additional documents along with these documents to the embassy.
Invitation Letter: The official invitation letter is proof that your submitted paper and registration request are accepted by the conference board. It will be indicated in English and can help you with your visa application.
* Only registered participants will receive an official invitation letter
* Participants must complete registration and submit authenticated information to obtain an official invitation letter [i.e. passport (scanned copy), date of birth, mobile phone number, physical address, and a photograph].
*Contact the program manager for more information
*Official invitation letters will be provided only to attend the conference.
** IF YOUR APPLICATION IS TURN DOWN THEN THE ORGANIZING COMMITTEE OF Life Saving Drug-2023 CAN NOT CHANGE THE DECISION OF THE DEPARTMENT OF FOREIGN AFFAIRS AND WE WILL NOT HEAR ANY DISCUSSION OR CORRESPONDENCE WITH THE IND, MOFA OR EMBASSY ON BEHALF OF THE CANDIDATE **
Market Analysis
The lifesaving drug request is prognosticated to reach approx. USD billion by 2023 from USD billion in 2018, at an Emulsion Annual Growth Rate of6.4 during the prognosticated period. The Growth in the request of lifesaving drug is substantially directed by the increase in the frequency of habitual conditions, growth in technological advancements and the biologics request, and new product launches. On the other hand, the threat of needle stick injuries (numerous Infections) and the rise in pricing pressure are anticipated to claim request growth in the forthcoming times. In addition, the cases’ heartiness has progressed vastly with ever- perfecting robotization, and possibilities and capabilities of massive improvements in this field have pushed the medicine delivery assiduity to a new height, and in having an astonishing growth in medicine delivery systems for hormones and cardiology agents. The global request for advanced medicine delivery systems is prognosticated to quantum to a value of$196.4 billion by 2025 at a7.2 CAGR
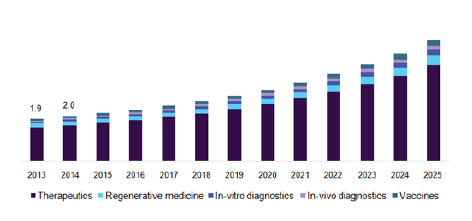
Pharmaceutical Sciences & Life Saving Drugs appertained to the phrasings, technologies, approaches, inventions and systems for transporting a pharmaceutical emulsion in the body as demanded to safely achieve its needed remedial effect. It is important to take a Regard on this content as it is directly related to one’s life. The request is presently estimated to be worth approx. USD18.5 billion for the time 2018 and is anticipated to reach USD51.7 billion by the end of 2025.
To Collaborate Scientific Professionals around the World
Conference Date August 28-29, 2023
For Sponsors & Exhibitors
Speaker Opportunity
Useful Links
Past Conference Report
Supported By
All accepted abstracts will be published in respective Conference Series International Journals.
Abstracts will be provided with Digital Object Identifier by








